Meet Our Board
Research Team
Outreach Team
Conference Team
Fundraising Team
Communications Team
Advocacy Team
Host a Fundraiser
Let's help one another find our way forward.
Our foundation advocates for people with Galactosemia and their families. We connect families and support networking efforts between clinicians and researchers.
Get Involved2026 Galactosemia Foundation Conference
Upcoming Event – July 16, 2026 - July 18, 2026
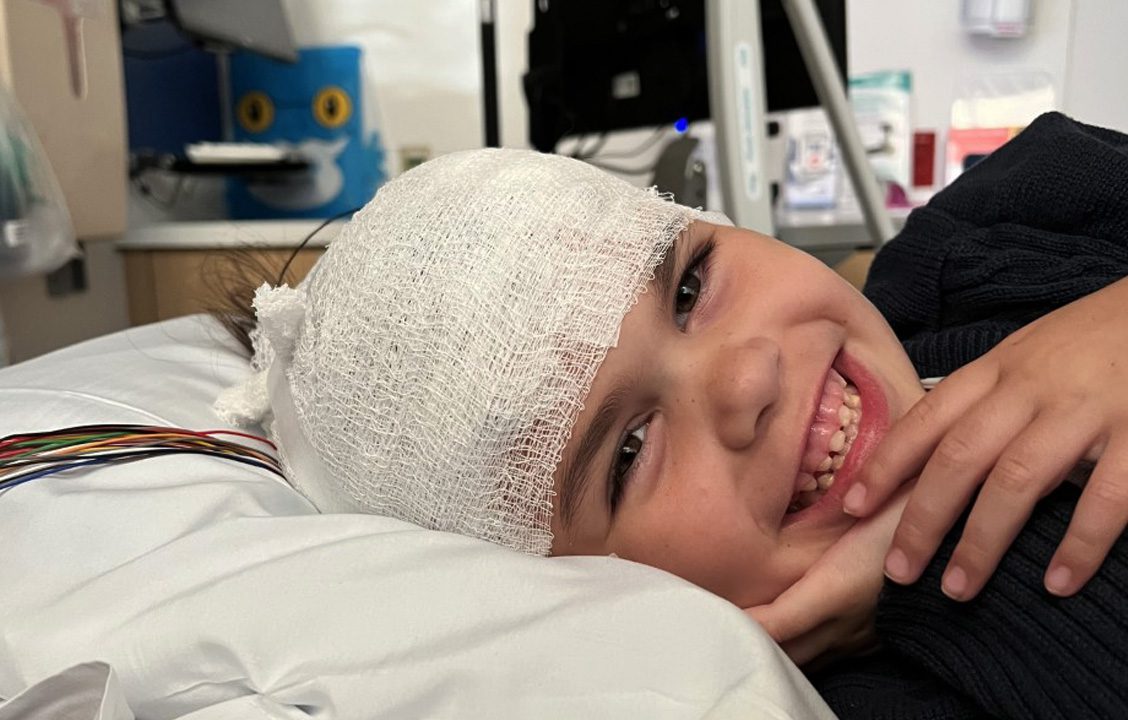
We Need You!
Advocating for a Treatment
The Galactosemia Foundation is urging the U.S. Food and Drug Administration (FDA) to incorporate the experiences and perspectives of people living with galactosemia in the agency’s review of govorestat (AT-007), the potential first-ever treatment for our rare genetic disease.