Meet Our Board
Research Team
Outreach Team
Conference Team
Fundraising Team
Communications Team
Advocacy Team
Host a Fundraiser
Stay Informed
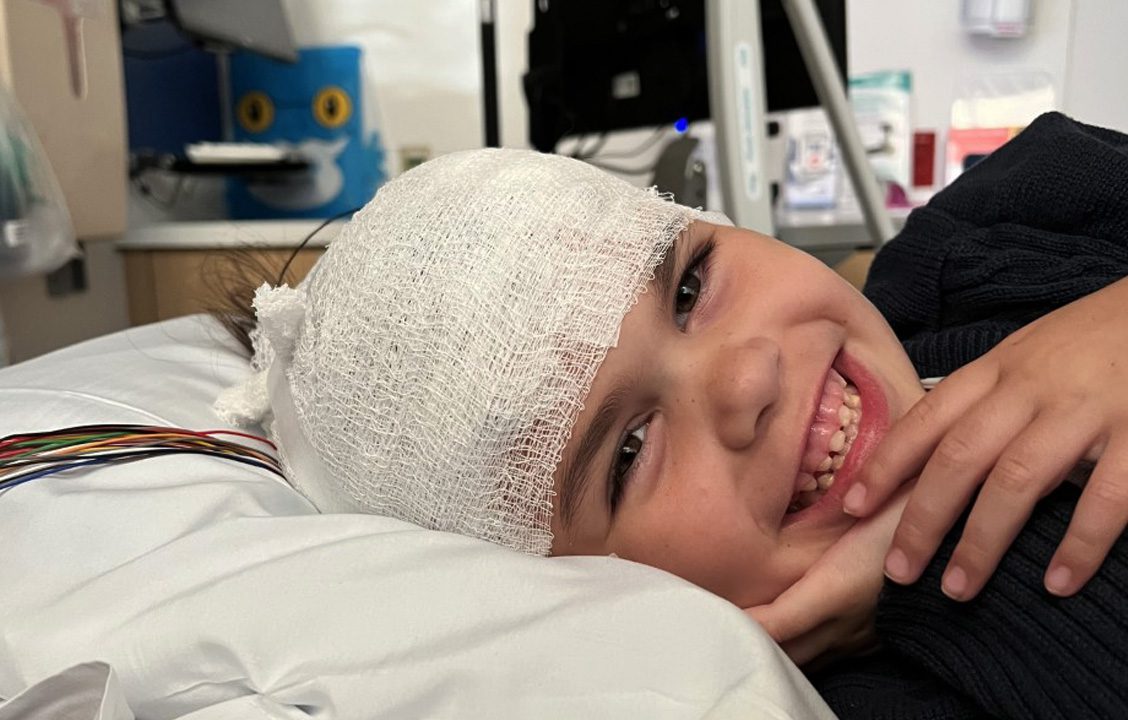
We Need You!
Advocating for a Treatment
The Galactosemia Foundation is urging the U.S. Food and Drug Administration (FDA) to incorporate the experiences and perspectives of people living with galactosemia in the agency’s review of govorestat (AT-007), the potential first-ever treatment for our rare genetic disease.
Latest News
Funded Research
The Galactosemia Foundation provides funding for scientific and medical research related to the identification, treatment, and therapies related to classical galactosemia. The goal of this research initiative is to improve the lives of people with galactosemia.
Research In Progress
Many galactosemia research efforts are underway. Some of the studies are currently recruiting galactosemia community participants. Click on the information below to learn more about the various opportunities.